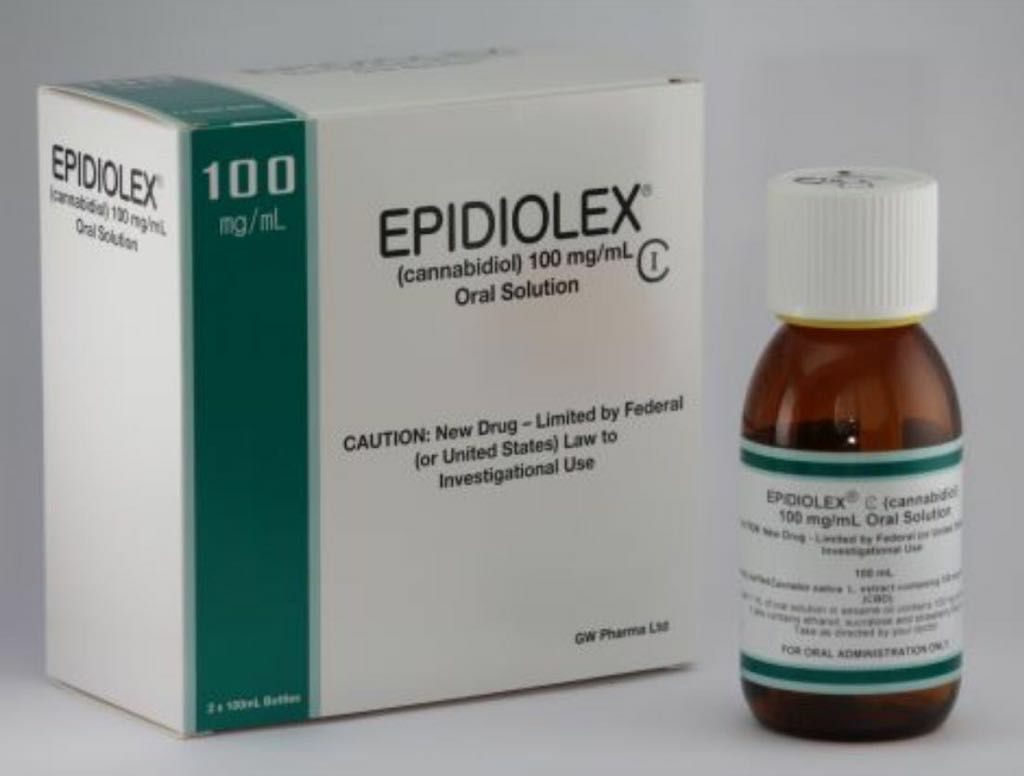
Epidiolex, a cannabis-based drug made up only of cannabidiol (CBD) has been approved by the Food and Drug Administration (FDA) in the United States of America (USA).
Em release, the US Food and Drug Administration confirmed approval of the first drug based on the species Cannabis Sativa L. Epidiolex is indicated for the control of seizures associated with epilepsy and stands out for the fact that CBD has no psychoactive properties and negligible side effects, having already been considered a “safe” substance by the World Health Organization (WHO).
Epidiolex is manufactured by the British pharmaceutical company GW Pharmaceuticals and has only CBD in its composition, a non-psychoactive component, being a purified form of one of the more than 80 cannabinoids present in the plant. The drug is manufactured by isolating high purity CBD crystals, then diluted in sesame seed oil.
Purified plant-derived CBD formulation approved
Epidiolex has as active ingredient the cannabidiol while the excipients described in the document are dehydrated alcohol, sesame seed oil, strawberry flavor and sucralose. Further information about this medicine is at FDA approved leaflet and written by Greenwich Biosciences Inc., a US subsidiary of GW Pharmaceuticals, which will market Epidiolex in the US.

Cannabis Sativa L. plants produced by GW Pharmaceuticals, from which CBD is extracted to produce Epidiolex – Photo: GW Pharmaceuticals
Epidiolex is approved for patients aged 2 years and older and for two types of epileptic syndromes: Dravet syndrome, a rare genetic disorder of the brain that begins in the first year of life, and Lennox-Gastaut syndrome, epilepsy with multiple types of seizures that begin in early childhood, usually between the ages of three and five. The medicine consists of an oral solution that is administered in the form of drops.
"This is important medical progress," said FDA Commissioner Scott Gottlieb. “Because of the adequate and well-controlled clinical studies that supported this approval, prescribing physicians can have confidence in the uniformity and assurance of drug delivery.”
Epidiolex is the “first purified pharmaceutical grade formulation based on cannabidiol extracted from cannabis plants, the non-psychoactive cannabinoid in cannabis, and the first to enter a new category of anti-epileptic drugs,” according to the release made public by the biopharmaceutical company that manufactures and markets Epidiolex, GW Pharmaceuticals.
Epidiolex only arrives in Europe next year
The FDA approval of this drug limits its use to the US territory, but the European Medicines Agency is also reviewing the Marketing Authorization (MA) application for Epidiolex. However, the approval decision in Europe is only expected to be announced in the first quarter of 2019, according to a company press release in February.
In the meantime, it is possible that, once on the market, Epidiolex could be prescribed for conditions other than those for which it was approved. This type of use is called use. off-label medical prescription and is a common practice with many drugs.

Dravet Portugal Association website – www.dravet.pt
Dravet Portugal can benefit a lot from Epidiolex
In statements to Cannapress, the Dravet Portugal Association welcomes the “very important step taken by the FDA with this approval”. The market introduction of this drug “has been studied for various types of epilepsies” argued Victor Mateus, Vice-President of Dravet Portugal, concluding that it “brings some hope to many families”.
Currently, Portuguese families with children affected by Dravet Syndrome and Lennox-Gastaut Syndrome do not have a safe source of CBD oils, and the authorities have even seized several CBD-based products for patients at Customs. According to the legislation in force, the same oils are marketed as food supplements and are not legally recognized as medicines or treatments for any clinical condition.
____________________________________________________________
Featured Photo: DR