the canadian company Canopy Growth, one of the international cannabis giants, has filed a lawsuit against the GW Pharmaceuticals PLC, from the United Kingdom, on the 22nd of December. The Canadian company alleges infringement of intellectual property in the preparation of extracts by the method of extraction with Carbon Dioxide (CO2), according to the Forbes.
Christmas 2020 is marked by the legal action brought by the company Canopy Growth against the first company with a drug approved by the American and European health regulators (the FDA e EMA, respectively), marking the beginning of a legal battle over intellectual property. At issue are the techniques for extracting cannabinoids from the cannabis plant. The action was known on December 22 and is filed in the District Court for the Western District of Texas, USA. The document alleges that GW Pharma's anticonvulsant drug, Epidiolex, is produced using Canopy's patented extraction process.
The lawsuit refers to the “continuous and unauthorized use” of Canopy's patented processes to extract cannabidiol or CBD from cannabis plant material by GW Pharma. CBD is a cannabinoid that is naturally present in cannabis, like many other cannabinoids. It is used as the API (Pharmacologically Active Ingredient) of the cannabis plant and is often used for medicinal purposes. CBD does not have the toxic effects caused by tetrahydrocannabinol (THC), another of the active ingredients in cannabis.
Patent 632 was allegedly used by GW without authorization

Apeks CO2 extraction machine on the Future 4200 website
According to Canopy, Patent No. 10.870.632 or the “632 Patent” was duly and legally issued by the United States Patent and Trademark Office (USPTO) and the company acquired all right, title and interest in the '632 patent after its 2019 acquisition by C3 Cannabinoid Compound Company of Germany, founded by herbal medicine manufacturer Bionorica SE.
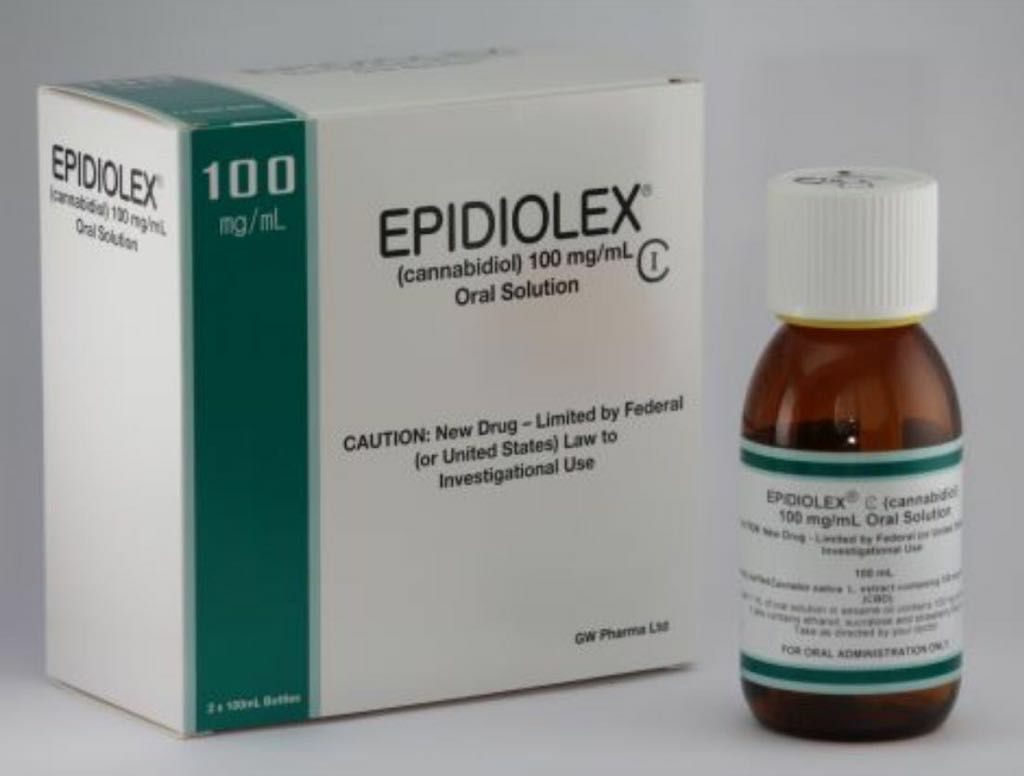
Canopy Growth claimed that GW Pharma manufactures the active pharmaceutical ingredient (API) for the manufacture of Epidiolex, a CBD-rich drug, using the CO-based extraction process.2 described and claimed in the '632 Patent.
GW Pharma's oral pharmaceutical formulation of purified cannabidiol is already approved in the US and Europe for the treatment of seizures associated with Lennox-Gastaut syndrome (LGS) or Dravet syndrome, two rare conditions of childhood epilepsy, typically characterized by being refractory. , that is, they do not respond correctly to conventional medicines used in epilepsy. In Europe, the drug is approved under the trade name EPIDYOLEX (EPIDIOLEX in the United States of America) for the treatment of seizures associated with LGS or Dravet syndrome.
Canopy Growth alleged in the lawsuit that GW Pharma has been monitoring the '632 family of patents for more than fourteen years, having refused to license the parent patent in 2017.
“This case is not about restricting patient access to Epidiolex. Instead, Canopy brings this action to put an end to GW's knowledge and unauthorized use of our company's intellectual property."
Canopy Growth will demand damages from the UK company to compensate it for the breach of patent by GW Pharma.
Read the process text below:
489007576-Canopy-vs-GW-CBD-Extraction-Lawsuit-and-Patent
________________________________________________________________________