O infarmed — National Authority for Medicines and Health Products IP granted, by the end of June 2022, 61 licenses and 172 pre-licenses to companies with activities related to medical cannabis. The report was disclosed yesterday first hand to Cannareporter and demonstrates, unequivocally, the attractiveness of the cannabis sector in Portugal, with several licensing applications under analysis. according to the data, the number of companies could double in the coming years.
Portugal started in 2018 the regulation of cannabis for medicinal purposes, including legal provisions for cannabis production and processing activities, intended for the medical market. Despite the interest of companies and Portugal having issued its first license in 2014, it was with the progressive clarification of the legal framework that the volume of licenses soared.
According to the report published by the Inspection and Licensing Directorate of Infarmed, to which Cannareporter had access yesterday, first hand, the Portuguese medical cannabis industry has been consolidating in Portugal and the number of licensed companies can more than what to duplicate.
Licenses, Pre-Licenses and Authorizations
The report published by the Portuguese regulator takes stock of the evolution of activities related to the field of cannabis for medicinal purposes in Portugal, with data up to June 2022. The document describes the indicators relating to the number of entities and the number global ordering and encompasses the activities of Cultivation, Manufacturing, Import/Export and Marketing.
According to data from Infarmed, 61 licenses have been granted to date, 20 of which are for cultivation, 8 for processing, 23 for import/export and 10 for marketing.
Import and export skyrocket
The signs of vitality of the cannabis industry in Portugal are attested by the more than 100 entities that have already started the licensing process.
According to the report, in June there were 40 entities in the first phase of licensing (Without documentary suitability - requests that did not present all the necessary documentation for the continuity of the process), and 90 in the second phase (With documentary suitability - after issuance of pre -licence, where the activity is initiated in a conditional and controlled manner).
The signs of maturation of the industry in European and global terms are evident in the data presented, where import/export activities are more expressive, with 93 entities requesting licensing from the regulator.
Among the 40 entities that are in the first phase of licensing, the activity that is most expressive is cultivation. The primary production activity has 33 entities in the first licensing stage. In the remaining activities, the entities in the first phase of the procedure are distributed among the activities of manufacturing (16 entities), import/export (14 entities) and commercialization, with 4 requests in the first phase of analysis. (See chart below)
172 pre-licenses granted to 90 entities
With regard to entities that are at a more advanced stage of licensing (second licensing phase), import/export and cultivation activities are the most expressive.
In total, Infarmed granted 172 pre-licenses, distributed among the activities of cultivation (55 licensing requests), import/export (79 licensing requests), manufacturing (24 licensing requests) and commercialization (10 licensing requests).( See chart below)
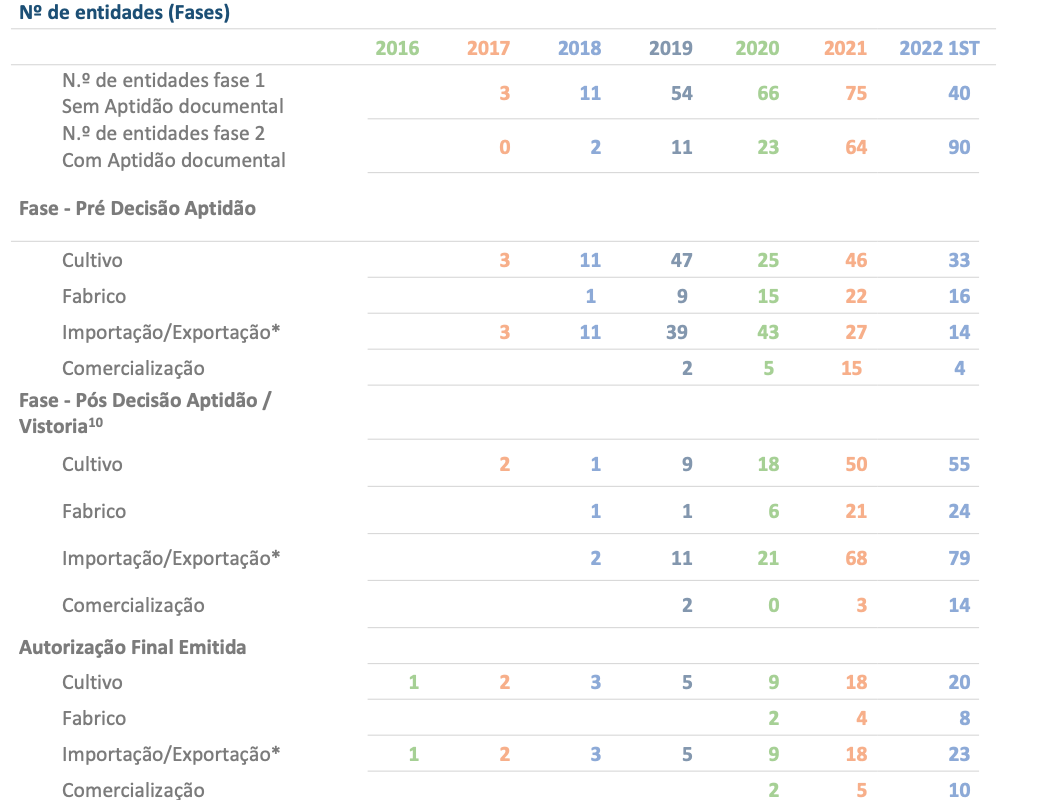
Number of entities with licensing requests in Portugal, distributed by the different licensing phases. Report “Medical Cannabis — Evolution and Activity”, published by Infarmed
If all the licensing requests under analysis were accepted, the number of entities operating in the national territory could exceed 150 companies.











































